Use the periodic table to write the electron configuration of selenium (Se).
...

Chemistry, 14.01.2021 21:40 mixedkiddo
Use the periodic table to write the electron configuration of selenium (Se).
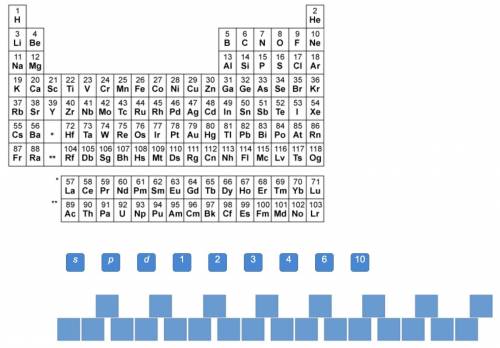

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:00
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
Questions







Biology, 11.06.2021 03:00


Mathematics, 11.06.2021 03:00


History, 11.06.2021 03:00

Mathematics, 11.06.2021 03:00

Mathematics, 11.06.2021 03:00

Mathematics, 11.06.2021 03:00

Mathematics, 11.06.2021 03:00


Mathematics, 11.06.2021 03:00





