Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1


Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3
You know the right answer?
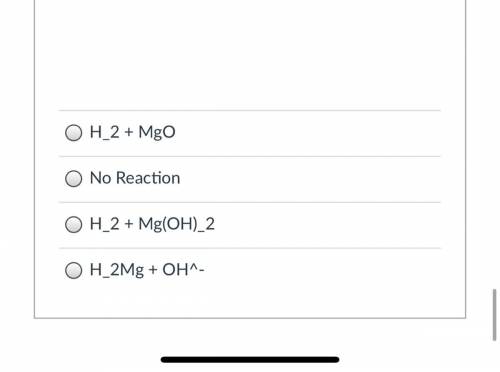
Predict the outcome of this reaction. Balancing doesn't matter.
Mg + H2O -->
...
Mg + H2O -->
...
Questions

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

History, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

History, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01