Chemistry, 14.01.2021 18:10 smallblueberry
i) Mass of CO2 formed = 2.241 g Molar mass of CO2 = Atomic mass of C + 2 (Atomic mass of O) = 12 + 2 (16) g/mol = 44 g/mol Thus, moles of carbon dioxide formed

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3
You know the right answer?
i) Mass of CO2 formed = 2.241 g Molar mass of CO2 = Atomic mass of C + 2 (Atomic mass of O) = 12 + 2...
Questions


Social Studies, 21.01.2021 23:30



Mathematics, 21.01.2021 23:30

Mathematics, 21.01.2021 23:30

Social Studies, 21.01.2021 23:30

Mathematics, 21.01.2021 23:30

Mathematics, 21.01.2021 23:30


Biology, 21.01.2021 23:30



Mathematics, 21.01.2021 23:30

Mathematics, 21.01.2021 23:30


Mathematics, 21.01.2021 23:30

Mathematics, 21.01.2021 23:30


Mathematics, 21.01.2021 23:30

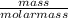
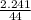 = 0.05mole
= 0.05mole