
6. How many grams are in 3.45 x1022 molecules of Na2CO3?
7. How many moles are in 26.88g of KCI?
8. How many moles are in 0.02345 g of NH4C2H302?
9. How many grams are in 8.64x1025 molecules of KCI?
10. How many moles are in 3.45 g of Pb(Cr2O7) ?
11. How many molecules are in 0.00487 g of KCI?
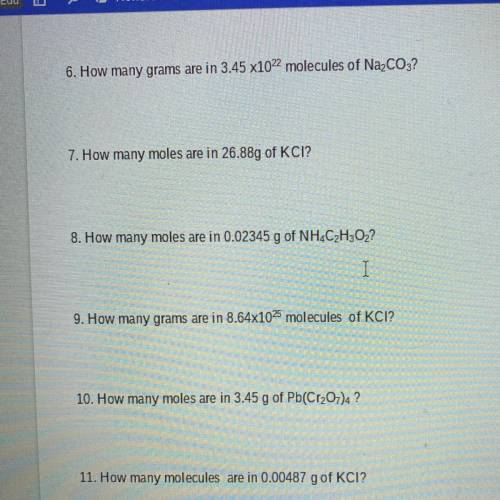

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1


Chemistry, 22.06.2019 14:30
Select the word from the list that best fits the definition the nuclear family into which a person is born or adopted.
Answers: 2

Chemistry, 23.06.2019 07:00
Why do the strengths of london (dispersion) forces generally increase with increasing molecular size? choose one: a. heavier atoms have stronger attractions for each other than lighter atoms. b. dispersion forces are all equal in magnitude; there is no size dependence. c. dispersion forces arise from the attraction between the nuclei of atoms, and larger molecules have larger nuclei. d. dispersion forces arise from dipoles caused by the electron distribution being distorted. larger molecules have more electrons and, therefore, more distortions and a bigger force. e. dispersion forces depend on distance. larger molecules are farther apart and so the forces are smaller.
Answers: 2
You know the right answer?
6. How many grams are in 3.45 x1022 molecules of Na2CO3?
7. How many moles are in 26.88g of KCI?
Questions


Mathematics, 17.03.2020 23:32


English, 17.03.2020 23:32



Mathematics, 17.03.2020 23:32


History, 17.03.2020 23:32


History, 17.03.2020 23:33

English, 17.03.2020 23:33


Mathematics, 17.03.2020 23:33

Mathematics, 17.03.2020 23:33


Mathematics, 17.03.2020 23:33

Mathematics, 17.03.2020 23:33

Mathematics, 17.03.2020 23:33




