
Chemistry, 14.01.2021 02:20 gilliancarter2202
Analyze the reaction to determine whether the reaction is exothermic or endothermic. Explain your reasoning.
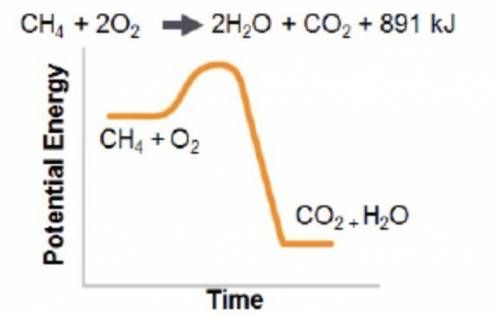

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 21.06.2019 22:20
One or more substances changing into one or more substances is an example of a
Answers: 1

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2
You know the right answer?
Analyze the reaction to determine whether the reaction is exothermic or endothermic. Explain your re...
Questions



Mathematics, 20.08.2020 06:01

Mathematics, 20.08.2020 06:01


Advanced Placement (AP), 20.08.2020 06:01




History, 20.08.2020 06:01


English, 20.08.2020 06:01




History, 20.08.2020 06:01

Mathematics, 20.08.2020 06:01

History, 20.08.2020 06:01




