
Chemistry, 14.01.2021 01:00 imorn20731
2Na + Cl2 → 2NaCl
How many moles of Cl2 do you need to use in order to react with 34.485 g of Na?
moles (put 1 zero in front of the decimal)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3


Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1
You know the right answer?
2Na + Cl2 → 2NaCl
How many moles of Cl2 do you need to use in order to react with 34.485 g of Na?
Questions






Advanced Placement (AP), 19.09.2020 01:01




English, 19.09.2020 01:01



History, 19.09.2020 01:01

History, 19.09.2020 01:01


History, 19.09.2020 01:01

History, 19.09.2020 01:01

Mathematics, 19.09.2020 01:01

Mathematics, 19.09.2020 01:01

English, 19.09.2020 01:01

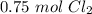
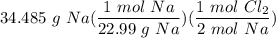 Multiply/Divide:
Multiply/Divide: 

