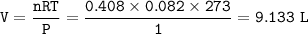Chemistry, 13.01.2021 07:10 tawna6988owtjg6
Trimethylamine, (CH3)2N is a weak base (K6 = 6.3 x 10-5). What volume of this gas, measured at STP, must be dissolved in 2.5 L of solution to give that solution a pOH of 2.50? PLEASE HELP ITS DUE IN 20 mins :

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 22.06.2019 23:00
Which type of intermolecular attractions holds ammonia molecules together with other ammonia molecules?
Answers: 3

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3

Chemistry, 23.06.2019 10:20
El amoniaco y el fluor reaccionan para formar tetrafluoruro de dinitrogeno y fluoruro de hidrogeno. segun la reaccion: nh3 + f2 ⇒ n2f4 + hf si reaccionan 5 gramos de amoniaco y 20 gramos de fuor, ¿cuantos gramos de fluoruro de hidrogeno se producen?
Answers: 2
You know the right answer?
Trimethylamine, (CH3)2N is a weak base (K6 = 6.3 x 10-5). What volume of this gas, measured at STP,...
Questions

Mathematics, 16.09.2019 18:50





Mathematics, 16.09.2019 19:00


Biology, 16.09.2019 19:00


Chemistry, 16.09.2019 19:00


History, 16.09.2019 19:00


Mathematics, 16.09.2019 19:00

Mathematics, 16.09.2019 19:00

English, 16.09.2019 19:00

Social Studies, 16.09.2019 19:00



![\tt [OH^-]=10^{-2.5}=0.0032=3.2\times 10^{-3}](/tpl/images/1031/5783/b6571.png)
![\tt [OH^-]=\sqrt{Kb.M}\\\\(3.2\times 10^{-3})^2=6.3\times 10^{-5}\times M\\\\M=0.163](/tpl/images/1031/5783/8f3e1.png)