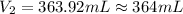Chemistry, 11.01.2021 16:10 makailaaa2
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do this by pouring out some 1.58M aqueous sodium carbonate stock solution into a graduated cylinder and diluting it with distilled water. Calculate the volume in mL of the sodium carbonate stock solution that the chemist should pour out. Round your answer to 3 significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Write two balanced equations 1. dissolving of solid sodium hydroxide in water 2. the reaction of sodium hydroxide solution with hydrochloric acid
Answers: 1

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2

Chemistry, 22.06.2019 08:30
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2
You know the right answer?
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do th...
Questions


Geography, 28.09.2021 15:20

Mathematics, 28.09.2021 15:20


English, 28.09.2021 15:20


Chemistry, 28.09.2021 15:20




Social Studies, 28.09.2021 15:20


Mathematics, 28.09.2021 15:20



Mathematics, 28.09.2021 15:20

Mathematics, 28.09.2021 15:20

English, 28.09.2021 15:20


Physics, 28.09.2021 15:20

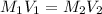
 = molarity of aqueous sodium carbonate
= molarity of aqueous sodium carbonate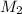 = molarity of aqueous sodium carbonate stock solution
= molarity of aqueous sodium carbonate stock solution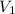 = volume of aqueous sodium carbonate
= volume of aqueous sodium carbonate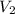 = volume of aqueous sodium carbonate stock solution
= volume of aqueous sodium carbonate stock solution