
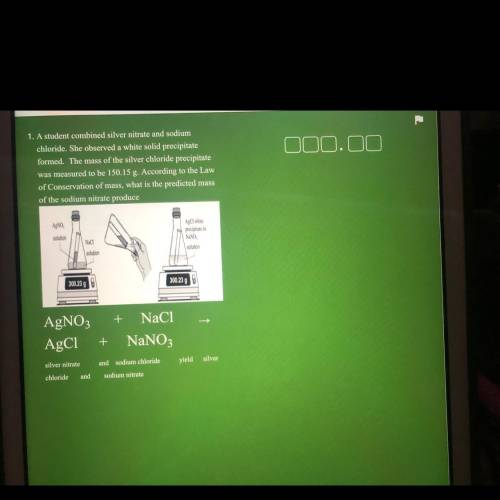
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
formed. The mass of the silver chloride precipitate
was measured to be 150.15 g. According to the Law
of Conservation of mass, what is the predicted mass
of the sodium nitrate produce
AgNO
AgCl white
precipitate in
solution
Naci
NaNO
solution
solution
300.23 g
300.23 90
+ NaCl
AgNO3
AgC1
+
NaNO3
silver nitrate
and sodium chloride
yield silver
chloride
and
sodium nitrate

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:50
Select the correct answer how does the heat content of the reaction change in the process of photosynthesis when a glucose molecule is formed? ca the value of is negative the value of qis positive the value of a remains constant the value of a decreases the value of equals zero e
Answers: 2

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1


Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
Questions

Mathematics, 08.12.2020 01:30


Mathematics, 08.12.2020 01:30

Business, 08.12.2020 01:30

Social Studies, 08.12.2020 01:30


Mathematics, 08.12.2020 01:30




Mathematics, 08.12.2020 01:30

Mathematics, 08.12.2020 01:30



Chemistry, 08.12.2020 01:30

Mathematics, 08.12.2020 01:30


Mathematics, 08.12.2020 01:30

Social Studies, 08.12.2020 01:30

World Languages, 08.12.2020 01:30



