
Chemistry, 08.01.2021 20:10 kinderc9330
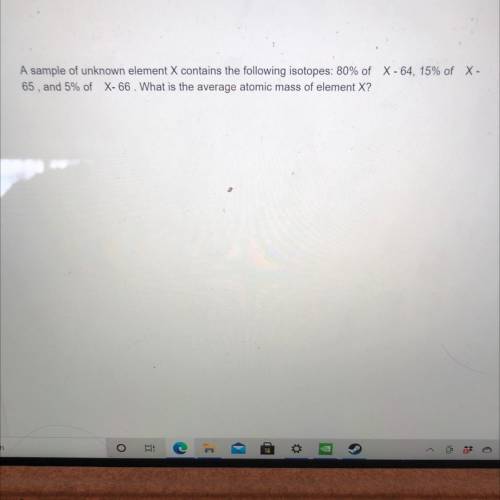
A sample of unknown element X contains the following isotopes: 80% of C -64, 15% of X - 65, and 5% of X-66. What is the average atomic mass of element X? Can somebody help me with this question. Will mark brainliest.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 05:30
Which of the following two events occur to create a sea breeze? select all that apply. warm air rises on the ocean and moves toward the land to cool warm air rises on land and moves toward the ocean to cool cool air moves from the ocean to be warmed by the land cool air moves from the land to be warmed by the ocean
Answers: 3

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 23.06.2019 05:40
Convert a speed of 201 cm/s to units of inches per minute. also, show the unit analysis by dragging components into the unit‑factor slots.
Answers: 1
You know the right answer?
A sample of unknown element X contains the following isotopes: 80% of C -64, 15% of X - 65, and 5% o...
Questions

Biology, 25.08.2019 04:50




Social Studies, 25.08.2019 04:50

Health, 25.08.2019 04:50


Mathematics, 25.08.2019 04:50


Mathematics, 25.08.2019 04:50


English, 25.08.2019 04:50

Biology, 25.08.2019 04:50


Social Studies, 25.08.2019 04:50

Mathematics, 25.08.2019 04:50

English, 25.08.2019 04:50



Mathematics, 25.08.2019 04:50



