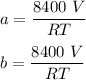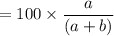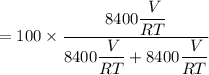Chemistry, 07.01.2021 17:30 dawsgreer4302
A mixture of ethyne gas (C2H2) and methane gas (CH4) occupied a certain volume at a total pressure of 16.8 kPa. When the sample burned, the products were CO2 gas and H2O vapor. The CO2 was collected and its pressure found to be 25.2 kPa in the same volume and at the same temperature as the original mixture. What percentage of the original mixture was methane

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:10
Which form of relativism states that people rely on their own standards of right and wrong when making a decision?
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
A mixture of ethyne gas (C2H2) and methane gas (CH4) occupied a certain volume at a total pressure o...
Questions

Health, 14.09.2021 04:20


Mathematics, 14.09.2021 04:20

Mathematics, 14.09.2021 04:20

Chemistry, 14.09.2021 04:20

Mathematics, 14.09.2021 04:20



Mathematics, 14.09.2021 04:20



Mathematics, 14.09.2021 04:20


English, 14.09.2021 04:20

English, 14.09.2021 04:20

Mathematics, 14.09.2021 04:20



Computers and Technology, 14.09.2021 04:20

Chemistry, 14.09.2021 04:20

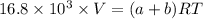
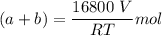 --- (1)
--- (1)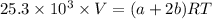
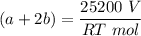 ---- (2)
---- (2)