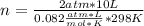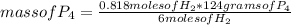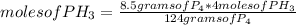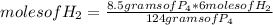Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Diffraction is when light is bent around obstructions. which of the these observation about clouds would indicate diffraction? a) after rain storms, you can sometimes see rainbows. b) clouds are white or gray and cannot be seen through. c) on a cloudy day, the temperature tends to be cooler than a sunny day. d) the edges of dark clouds appear lighter. this
Answers: 3

Chemistry, 22.06.2019 14:00
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2

Chemistry, 22.06.2019 14:20
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d.lytic
Answers: 1

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
If 8.50 g of phosphorus reacts with hydrogen gas at 2.00 atm in a 10.0-L container at 298 K, calcula...
Questions

Mathematics, 03.02.2021 01:10


History, 03.02.2021 01:10

English, 03.02.2021 01:10

English, 03.02.2021 01:10

Spanish, 03.02.2021 01:10

Chemistry, 03.02.2021 01:10



Mathematics, 03.02.2021 01:10



English, 03.02.2021 01:10



Chemistry, 03.02.2021 01:10




Chemistry, 03.02.2021 01:10

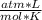 T= 298 K
T= 298 K