HELP
10 PTS!!
the answers are given already but my teacher wants me to show work...pls help!!...

Chemistry, 07.01.2021 09:40 JellalFernandes
HELP
10 PTS!!
the answers are given already but my teacher wants me to show work...pls help!!!


Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:04
4moles of nitrogen gas are confined to a 6.0 l vessel at 177 °c and 12.0 atm. if the vessel is allowed to expand isothermally to 36.0 l, what would be the final pressure?
Answers: 3

Chemistry, 21.06.2019 18:10
Using complete sentences, explain how to predict the products and balance the reaction between sulfuric acid and potassium hydroxide.
Answers: 1

Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1

Chemistry, 22.06.2019 00:30
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3
You know the right answer?
Questions





Health, 21.03.2020 02:44




Mathematics, 21.03.2020 02:44







Mathematics, 21.03.2020 02:45




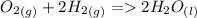
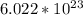 to get the number of molecules. We get: 2.41 * 10^24 molecules of water.
to get the number of molecules. We get: 2.41 * 10^24 molecules of water. 

