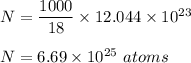Chemistry, 07.01.2021 05:20 walkerobrien5
18.0 mL of water contains 6.022 x 1023 water molecules. How many hydrogen atoms are in 1.00 L of water? (Each water molecule, H2O, contains two hydrogen atoms.)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Which mathematical relationship should you us to convert moles of a substance into grams
Answers: 1

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
18.0 mL of water contains 6.022 x 1023 water molecules. How many hydrogen atoms are in 1.00 L of wat...
Questions



Mathematics, 06.12.2019 07:31

English, 06.12.2019 07:31



Mathematics, 06.12.2019 07:31

Social Studies, 06.12.2019 07:31


Mathematics, 06.12.2019 07:31

Mathematics, 06.12.2019 07:31

Mathematics, 06.12.2019 07:31

English, 06.12.2019 07:31

English, 06.12.2019 07:31



Mathematics, 06.12.2019 07:31



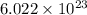 water molecules.
water molecules.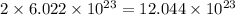 .
.