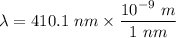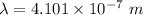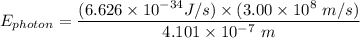The wavelength of the violet light emitted from a hydrogen atom is 410.1 nm. This light is a result of electronic transitions between the n = 5 and n = 2 energy levels. How much higher in energy is the n = 5 energy level than the n = 2 energy level?
Select one:
a. 3.000 x 108 J
b. 1.114 x 10-14 J
c. 2.436 x 10-18 J
d. 1.616 x 10-36 J
e. 4.847 x 10-19 J

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
An object becomes electrically charged when: electrons are created in it electrons from it are destroyed electrons are transferred to it protons from it are destroyed protons are created in it
Answers: 1


Chemistry, 23.06.2019 06:00
•what conclusions can you make about the relationship between the volume of a gas and its temperature? • what conclusions can you make about the relationship between the volume of a gas and its pressure? • what possible variables have you not accounted for? as you did the procedures, is it possible that the atmospheric pressure may have changed? if it did change over the course of your experiment, then how would your results have been affected?
Answers: 3

Chemistry, 23.06.2019 12:30
How do you interpret a chromatogram for what mixtures contain?
Answers: 3
You know the right answer?
The wavelength of the violet light emitted from a hydrogen atom is 410.1 nm. This light is a result...
Questions


Mathematics, 10.07.2019 16:20

Mathematics, 10.07.2019 16:20

Mathematics, 10.07.2019 16:20









Biology, 10.07.2019 16:30



Health, 10.07.2019 16:30


History, 10.07.2019 16:30

Business, 10.07.2019 16:30

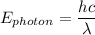
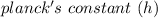 = 6.626 * 10 ^{-34} J.s
= 6.626 * 10 ^{-34} J.s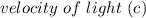 = 3.00 * 10^8 m/s
= 3.00 * 10^8 m/s