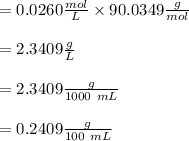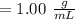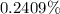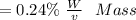Chemistry, 04.01.2021 02:00 vazquezemmy8
A sample of oxalic acid is titrated with a standardized solution of KMNO4. A 25 mL sample of oxalic acid required 12.7 mL of 0.0206 M KMnO4 to achieve a pink colored solution. The balanced equation for this reaction is shown below:
6 H+ (aq) + 2 MnO4 - (aq) + 5 H2C2O4(aq) → 10 CO2(g) +8 H2O(l) + 2Mn2+(aq)
Required:
a. What does the pink color signify in this reaction?
b. What is the ratio of MnO4 - ions to H2C2O4 molecules in this reaction?
c. How many moles of MnO4 - ions reacted with the given amount of oxalic acid solution?
d. How many moles of H2C2O4 were present?
e. What was the molarity of the oxalic acid solution?
f. If the density of the oxalic acid solution was 1.00 g/mL, what was the percentage by mass of oxalic acid in the solution?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:00
When a comet collides with earth, it adds material to our planet and causes great damage. therefore, a collision like this is a a. destructive force b. constructive force c. geologic process and event d. constructive and destructive force
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1


You know the right answer?
A sample of oxalic acid is titrated with a standardized solution of KMNO4. A 25 mL sample of oxalic...
Questions

Biology, 25.01.2020 20:31




Physics, 25.01.2020 20:31


History, 25.01.2020 20:31

Spanish, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

Biology, 25.01.2020 20:31




Mathematics, 25.01.2020 20:31


Health, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

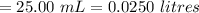
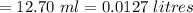
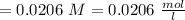
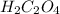 molecules mole ratio to
molecules mole ratio to 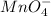 ions:
ions: 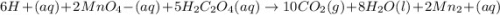
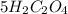 : :
: : 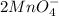
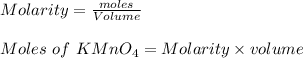
 mole ratio
mole ratio 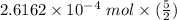
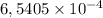 mol are present in the sample
mol are present in the sample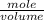
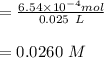
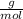 .
.