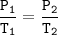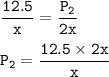Chemistry, 02.01.2021 20:10 kaylallangari1509
Without completing the calculations, determine what the new pressure will be in the problem below. Also,
explain how you were able to determine the new pressure without completing the calculations. Pay special
attention to how the temperature is changing in the problem.
A sample of ozone (0:2) is stored at x kelvin and 12.5 Pa. If the temperature is doubled to 2x kelvin,
what will the new
pressure be?

Answers: 1


Another question on Chemistry


Chemistry, 21.06.2019 17:00
What mass of carbon dioxide is produced from the complete combustion of 4.50×10−3 g of methane?
Answers: 2


Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
You know the right answer?
Without completing the calculations, determine what the new pressure will be in the problem below. A...
Questions



Health, 04.07.2019 22:00




Social Studies, 04.07.2019 22:00

English, 04.07.2019 22:00

Biology, 04.07.2019 22:00

History, 04.07.2019 22:00

Social Studies, 04.07.2019 22:00



History, 04.07.2019 22:00

Social Studies, 04.07.2019 22:00





Mathematics, 04.07.2019 22:00