
Chemistry, 31.12.2020 01:00 maddie53116
Use your knowledge of modern atomic theory and bonding to explain each of the following experimental observations. (Your explanations should be specific.) Refer to both substances when comparing. Within a family such as the alkaline earth metals, the ionic radius increases as the atomic number increases. The radius of the bromine atom is smaller than the radius of the bromide ion, Br-1. The first ionization energy of aluminum is lower than the first ionization energy of magnesium.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1
You know the right answer?
Use your knowledge of modern atomic theory and bonding to explain each of the following experimental...
Questions





Mathematics, 22.04.2020 07:01

SAT, 22.04.2020 07:01

Advanced Placement (AP), 22.04.2020 07:01



English, 22.04.2020 07:01

Arts, 22.04.2020 07:01


Mathematics, 22.04.2020 07:01






History, 22.04.2020 07:01

Biology, 22.04.2020 07:01

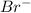 ion, the configuration of bromine earlier was
ion, the configuration of bromine earlier was 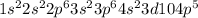 but on addition of the electron the p shell will now have 6 electrons resulting in
but on addition of the electron the p shell will now have 6 electrons resulting in 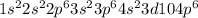 the newly added electron repels the former neutral orientation and as a result of repulsion the size increases.
the newly added electron repels the former neutral orientation and as a result of repulsion the size increases. 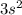 orbital whereas Aluminum has one electron in the 3p orbitals, fully filled 3s orbitals has strongly held by the nucleus in case of Mg whereas 3p orbitals is shielded from the nucleus therefore it becomes easy to take out 3p electron that 3s electron.
orbital whereas Aluminum has one electron in the 3p orbitals, fully filled 3s orbitals has strongly held by the nucleus in case of Mg whereas 3p orbitals is shielded from the nucleus therefore it becomes easy to take out 3p electron that 3s electron. 

