
Chemistry, 28.12.2020 18:00 davis52202
The solubility product for an insoluble salt with the formula MX4 is written as , where s is the molar solubility. Select one: a. Ksp

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Amass of 100.0 g of solute is dissolved in water so that 850. ml of a 0.7500 m solution has been prepared. what is the molar mass of the solute?
Answers: 2

Chemistry, 22.06.2019 21:00
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1

Chemistry, 23.06.2019 00:30
What is the chemical formula of magnesium bromide? a. mgbr2 b. mgbr c. mg2br2 d. mg2br
Answers: 3

Chemistry, 23.06.2019 04:10
Two solids are mixed in a flask and stirred. after a few minutes, the flask becomes cold. which of the following best describes this reaction? a. an exothermic reaction b. a combustion reaction c. an endothermic reaction d. a decomposition reaction
Answers: 1
You know the right answer?
The solubility product for an insoluble salt with the formula MX4 is written as , where s is the mol...
Questions





Mathematics, 04.04.2020 10:47







Biology, 04.04.2020 10:48



Computers and Technology, 04.04.2020 10:48


Mathematics, 04.04.2020 10:48



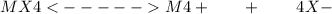
![Ksp = [M4+][X^-]^4](/tpl/images/1008/5610/d2716.png)
![Ksp = [s][4s]^4](/tpl/images/1008/5610/2aad8.png)


