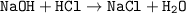Chemistry, 24.12.2020 21:20 issagirl05
During science lab, Jacob mixes together sodium hydroxide (NaOH) and hydrochloric acid (HCl). The two chemicals react to form sodium chloride (NaCl) and water (H2O).
Which of the following are reactants in the chemical reaction described above?
A.
hydrochloric acid and water
B.
sodium hydroxide and hydrochloric acid
C.
sodium hydroxide and sodium chloride
D.
sodium chloride and water

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 23.06.2019 00:30
Arrange the elements in order of increasing electronegativity. use the periodic table to you arrange the elements. p o k mg
Answers: 2
You know the right answer?
During science lab, Jacob mixes together sodium hydroxide (NaOH) and hydrochloric acid (HCl). The tw...
Questions

Biology, 19.07.2019 18:30

History, 19.07.2019 18:30

Business, 19.07.2019 18:30


Biology, 19.07.2019 18:30

Spanish, 19.07.2019 18:30

Social Studies, 19.07.2019 18:30

History, 19.07.2019 18:30

Mathematics, 19.07.2019 18:30

Social Studies, 19.07.2019 18:30

Advanced Placement (AP), 19.07.2019 18:30

Biology, 19.07.2019 18:30

Physics, 19.07.2019 18:30

History, 19.07.2019 18:30


History, 19.07.2019 18:30


Social Studies, 19.07.2019 18:30

Mathematics, 19.07.2019 18:30

History, 19.07.2019 18:30