
Calcium Carbonate reacts with dilute hydrochloric acid. The equation for the reaction is shown. CaCo3 + 2Hcl = Cacl2 + H2O + Co2 1 g of calcium Carbonate is added to 50cm3 of 0.05mol/dm3 hydrochloric acid. Which volume of carbon dioxide is made in this reaction?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Which of the following is not a good technique for managing used oil? a) have specific, labeled catch pans available for technicians who are collecting oil b) spills in your shop and any releases on pavement or outside should be poured down a drain c) do not use oil containers for antifreeze or other non-similar fluids d) be prepared for oil spills with the proper absorbents
Answers: 1

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1


You know the right answer?
Calcium Carbonate reacts with dilute hydrochloric acid. The equation for the reaction is shown. CaCo...
Questions

Biology, 25.11.2019 01:31

Mathematics, 25.11.2019 01:31



Mathematics, 25.11.2019 01:31




History, 25.11.2019 01:31

Biology, 25.11.2019 01:31

English, 25.11.2019 01:31



Engineering, 25.11.2019 02:31


History, 25.11.2019 02:31


Mathematics, 25.11.2019 02:31

English, 25.11.2019 02:31

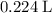 , assuming that this reaction took place under standard temperature and pressure, and that
, assuming that this reaction took place under standard temperature and pressure, and that 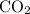 behaves like an ideal gas. Also assume that the reaction went to completion.
behaves like an ideal gas. Also assume that the reaction went to completion.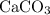 is the limiting reactant. How many moles of
is the limiting reactant. How many moles of 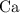 ,
,  , and
, and  on a modern periodic table:
on a modern periodic table: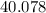 .
.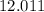 .
.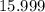 .
.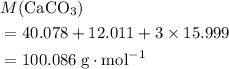 .
.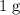 of
of 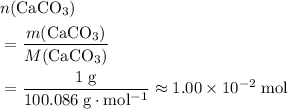 .
.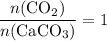 .
.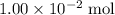 of
of 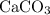 formula would be consumed. That would produce approximately
formula would be consumed. That would produce approximately 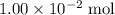 of
of 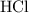 is the limiting reactant.
is the limiting reactant.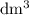 (so as to match the unit of concentration.)
(so as to match the unit of concentration.) .
.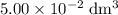 of this
of this 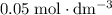
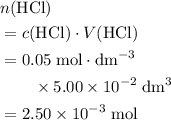 .
.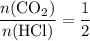 .
.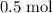 of
of 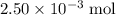 of
of 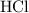 molecules would produce only one-half as many (that is,
molecules would produce only one-half as many (that is, 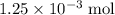 ) of
) of 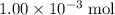 of
of 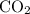 molecules would be produced.
molecules would be produced.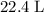 .
.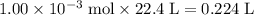 .
.

