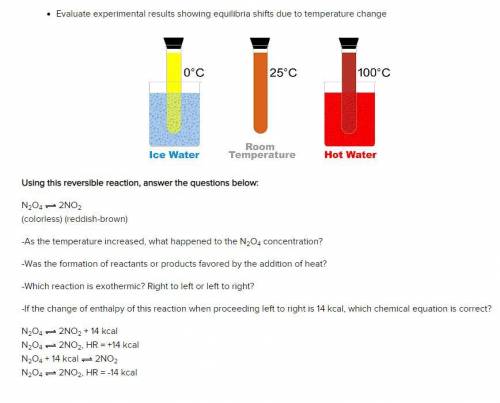
Using this reversible reaction, answer the questions below:
N2O4 2NO2
(colorless) (reddish-br...

Chemistry, 21.12.2020 14:00 mommer2019
Using this reversible reaction, answer the questions below:
N2O4 2NO2
(colorless) (reddish-brown)
-As the temperature increased, what happened to the N2O4 concentration?
-Was the formation of reactants or products favored by the addition of heat?
-Which reaction is exothermic? Right to left or left to right?
-If the change of enthalpy of this reaction when proceeding left to right is 14 kcal, which chemical equation is correct?
N2O4 2NO2 + 14 kcal
N2O4 2NO2, HR = +14 kcal
N2O4 + 14 kcal 2NO2
N2O4 2NO2, HR = -14 kcal

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1



Chemistry, 23.06.2019 05:00
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1
You know the right answer?
Questions

Chemistry, 14.09.2020 06:01

Geography, 14.09.2020 06:01

Mathematics, 14.09.2020 06:01

Mathematics, 14.09.2020 06:01

Chemistry, 14.09.2020 06:01

Mathematics, 14.09.2020 07:01

English, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01

Health, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01

English, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01

Health, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01

History, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01

Physics, 14.09.2020 07:01

Mathematics, 14.09.2020 07:01




