Chemistry, 19.12.2020 21:50 chriscendypierre56
An oxide of arsenic contains 3.26 g of arsenic and 1.04 g of oxygen. What is the empirical formula for this oxide?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 21.06.2019 18:30
Which orbitals form a pi bond? a.the s orbital and three p orbitals b.the s orbital and two p orbitals c.overlapping p orbitals d.overlapping hybrid orbitals
Answers: 2

Chemistry, 22.06.2019 15:30
The gulf stream is a warm water current that flows away from the equator to northern europe. witch of these does it cause. a. crashes of warm and cool water in the ocean b.colder climates near the equator c.large waves on the cost of europe d.warm climates in northern europe
Answers: 1

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2
You know the right answer?
An oxide of arsenic contains 3.26 g of arsenic and 1.04 g of oxygen. What is the empirical formula f...
Questions

History, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01


Biology, 05.10.2020 14:01








Mathematics, 05.10.2020 14:01


History, 05.10.2020 14:01


Computers and Technology, 05.10.2020 14:01

Biology, 05.10.2020 14:01


Physics, 05.10.2020 14:01

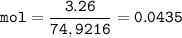
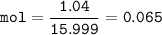 .
.