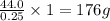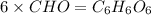Chemistry, 19.12.2020 16:10 amandaestevez030
Burning 12.00 g of an oxoacid produces 17.95 g of carbon dioxide and 4.87 g of water. Consider that 0.25 moles of oxoacid equals 44.0 g. For this compound, determine the empirical and molecular formula.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 23.06.2019 01:00
Which is true concerning the products and reactants of photosynthesis and cellular respiration? a. the products of photosynthesis are sugars and the reactants of cellular respiration are starches. b. the products of photosynthesis are reactants in cellular respiration. c. oxygen is needed for photosynthesis and is given off in cellular respiration.
Answers: 2
You know the right answer?
Burning 12.00 g of an oxoacid produces 17.95 g of carbon dioxide and 4.87 g of water. Consider that...
Questions


Mathematics, 26.05.2020 00:57

















Business, 26.05.2020 00:57

Computers and Technology, 26.05.2020 00:57

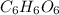
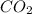 = 17.95 g
= 17.95 g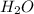 = 4.87 g
= 4.87 g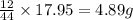 of carbon will be contained.
of carbon will be contained.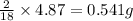 of hydrogen will be contained.
of hydrogen will be contained.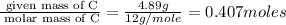
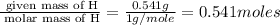



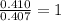
 .
.