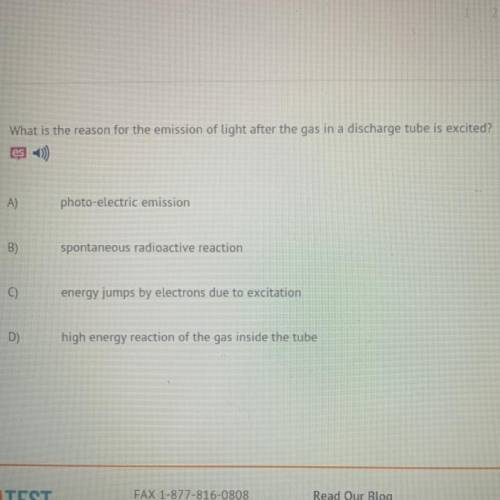
What is the reason for the emission of light after the gas in a discharge tube is excited
...

Chemistry, 18.12.2020 02:00 jinjermaxinejohnston
What is the reason for the emission of light after the gas in a discharge tube is excited

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
How does the principle of electromagnetism explain the interaction between earth’s magnetic field and the solar wind?
Answers: 1

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
Questions


History, 27.01.2020 20:31


Mathematics, 27.01.2020 20:31

Computers and Technology, 27.01.2020 20:31




Mathematics, 27.01.2020 20:31

Mathematics, 27.01.2020 20:31

History, 27.01.2020 20:31

History, 27.01.2020 20:31


History, 27.01.2020 20:31


Physics, 27.01.2020 20:31

History, 27.01.2020 20:31


Mathematics, 27.01.2020 20:31

Social Studies, 27.01.2020 20:31



