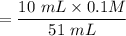Chemistry, 16.12.2020 16:50 canonmille2
10.0 mL of 0.0400 M KBrO3 and 10.0 mL 0.100 M HCl is mixed with 10.0 mL of 0.0100 M KI, 10.0 mL 0.00100 M Na2S2O3, 1.0 mL of starch solution, and 10.0 mL H2. What is the concentration of H in this mixture expressed as molarity?

Answers: 1


Another question on Chemistry


Chemistry, 21.06.2019 23:50
Why do scientists look for patterns in the world? a. patterns can explain observations. b. patterns never change, no matter what. c. patterns are easy for scientists to detect. d. patterns are all the same, through all time.
Answers: 1

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
10.0 mL of 0.0400 M KBrO3 and 10.0 mL 0.100 M HCl is mixed with 10.0 mL of 0.0100 M KI, 10.0 mL 0.00...
Questions

Health, 09.07.2019 02:00

Mathematics, 09.07.2019 02:00

Mathematics, 09.07.2019 02:00



Mathematics, 09.07.2019 02:00


Chemistry, 09.07.2019 02:00

World Languages, 09.07.2019 02:00


Social Studies, 09.07.2019 02:00




Mathematics, 09.07.2019 02:00

Mathematics, 09.07.2019 02:00


Mathematics, 09.07.2019 02:00


Mathematics, 09.07.2019 02:00

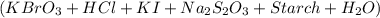
![[H^+] = \dfrac{ mL \ used \times 0.1 \ M}{total \ volume}](/tpl/images/0990/2194/57e25.png)