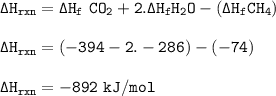Chemistry, 15.12.2020 15:00 lazerlemon500
The enthalpy change of formation of carbon dioxide is –394 kJ mol–1. The enthalpy change of formation of water is –286 kJ mol–1. The enthalpy change of formation of methane is –74 kJ mol–1. What is the enthalpy change of combustion of methane?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1

You know the right answer?
The enthalpy change of formation of carbon dioxide is –394 kJ mol–1. The enthalpy change of formatio...
Questions


Mathematics, 23.06.2019 15:00




Mathematics, 23.06.2019 15:00

Mathematics, 23.06.2019 15:00


Biology, 23.06.2019 15:00




Chemistry, 23.06.2019 15:00

Social Studies, 23.06.2019 15:00