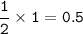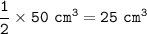4 Nitrogen monoxide reacts with oxygen like this:
2NO(gas) + O2(gas) -> 2NO2 (gas)
a. How many moles of oxygen molecules react
with I mole of nitrogen monoxide molecules?
b. What volume of oxygen will react with
50 cm of nitrogen monoxide?
c. Using the volumes in b, what is:
i. the total volume of the two reactants?
ii. the volume of nitrogen dioxide formed?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1

Chemistry, 23.06.2019 04:10
In an experiment, 45g of silicon tetrachloride are treated with 45ml of water. what is the theoretical yield in grams of hcl
Answers: 3

Chemistry, 23.06.2019 18:20
Sari is studying for different sample of materials. this table shows the mass and volume of the samples. which sample material(s) could be liquid
Answers: 3

Chemistry, 23.06.2019 18:40
Select the correct answer from each drop-down menu. in a reversible reaction, the forward reaction takes place the reverse reaction. such a reaction, on reaching equilibrium, will have .
Answers: 1
You know the right answer?
4 Nitrogen monoxide reacts with oxygen like this:
2NO(gas) + O2(gas) -> 2NO2 (gas)
a. How m...
a. How m...
Questions



History, 19.10.2019 08:50


Mathematics, 19.10.2019 08:50






Mathematics, 19.10.2019 08:50


English, 19.10.2019 08:50



Chemistry, 19.10.2019 08:50


History, 19.10.2019 08:50

SAT, 19.10.2019 08:50