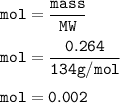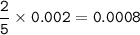Chemistry, 13.12.2020 08:20 barbie8775
0.2640 g of sodium oxalate is dissolved in a flask and requires 30.74 mL of potassium
permanganate (from a buret) to titrate it and cause it to turn pink (the end point).
The equation for this reaction is:
5Na2C2O4(aq) + 2KMnO4(aq) + 8H2SO4(aq) ---> 2MnSO4(aq) + K2SO4(aq) + 5Na2SO4(aq) +
10CO2(g) + 8H2O(1)
(A) How many moles of potassium permanganate have been titrated into the flask to reach the end point

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 14:30
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1

Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1
You know the right answer?
0.2640 g of sodium oxalate is dissolved in a flask and requires 30.74 mL of potassium
permanganate...
Questions

Mathematics, 27.02.2021 01:00

Mathematics, 27.02.2021 01:00

Mathematics, 27.02.2021 01:00



Mathematics, 27.02.2021 01:00

Mathematics, 27.02.2021 01:00

Chemistry, 27.02.2021 01:00

Social Studies, 27.02.2021 01:00

Mathematics, 27.02.2021 01:00


Mathematics, 27.02.2021 01:00

Mathematics, 27.02.2021 01:00


Mathematics, 27.02.2021 01:00

English, 27.02.2021 01:00


Mathematics, 27.02.2021 01:00


Geography, 27.02.2021 01:00