
Chemistry, 12.12.2020 15:50 nathancikra
5.00 ml of an H2SO4 solution with unknown molarity was diluted with 25.00 ml. This was tirated with 15.00ml of .1000 M NaOH to reach the equivalence point. What is the molarity of the original unknown H2SO4 solution? Also, what would the balanced chemical equation for this reaction be?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2


Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2
You know the right answer?
5.00 ml of an H2SO4 solution with unknown molarity was diluted with 25.00 ml. This was tirated with...
Questions

Mathematics, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01

Social Studies, 20.10.2020 21:01

Physics, 20.10.2020 21:01


Biology, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01

Computers and Technology, 20.10.2020 21:01



Mathematics, 20.10.2020 21:01

Social Studies, 20.10.2020 21:01



Mathematics, 20.10.2020 21:01

History, 20.10.2020 21:01




Social Studies, 20.10.2020 21:01

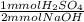 = 0.75 mmol H₂SO₄
= 0.75 mmol H₂SO₄

