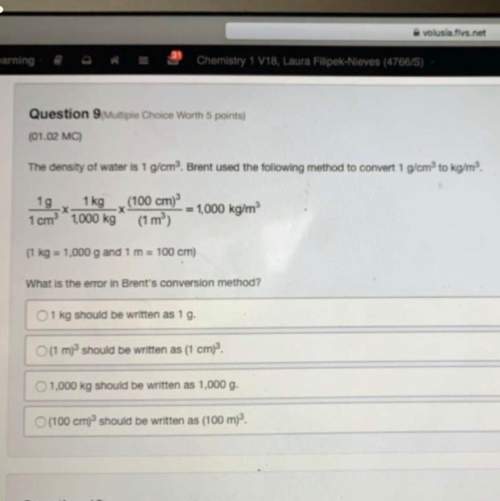
If the cell is initially at standard-state conditions, which of the following statements is true?
Zn2+(aq) + 2e- ↔Zn(s); E° = -0.76 V
Cu2+(aq) + 2e- ↔Cu(s); E° = 0.34 V
A. Initially Kc = 2 x 10-37, and it decreases with time.
B. Initially Kc = 2 x 1037, and it does not change with time.
C. Initially Kc = 2 x 1037, and it decreases with time.
D. Initially Kc = 2 x 1037, and it increases with time.
E. Initially Kc = 2 x 10-37, and it increases with time.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 16:40
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
If the cell is initially at standard-state conditions, which of the following statements is true?
Z...
Questions





Social Studies, 30.01.2020 07:05




Physics, 30.01.2020 07:05



Mathematics, 30.01.2020 07:43


Mathematics, 30.01.2020 07:43

History, 30.01.2020 07:43



Geography, 30.01.2020 07:43


Mathematics, 30.01.2020 07:43