Help please
If you have 98.078 grams of Sulfuric Acid, you have
mole(s) of Sulfuric Acid (H2S...

Chemistry, 08.12.2020 01:00 sfritz6456
Help please
If you have 98.078 grams of Sulfuric Acid, you have
mole(s) of Sulfuric Acid (H2SO4).

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 17:00
Complete each row of the table below by filling in the missing prefix or missing exponent.
Answers: 1


Chemistry, 23.06.2019 02:00
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3
You know the right answer?
Questions



Mathematics, 26.11.2019 21:31

Biology, 26.11.2019 21:31

Mathematics, 26.11.2019 21:31



Mathematics, 26.11.2019 21:31

English, 26.11.2019 21:31


Mathematics, 26.11.2019 21:31







Computers and Technology, 26.11.2019 21:31

Computers and Technology, 26.11.2019 21:31

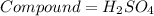
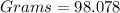
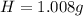 -- Hydrogen
-- Hydrogen 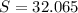 --- Sulfur
--- Sulfur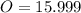 -- Oxygen
-- Oxygen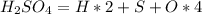
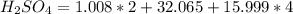
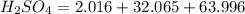
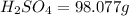
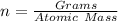
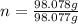
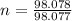
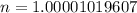
 (approximated).
(approximated).

