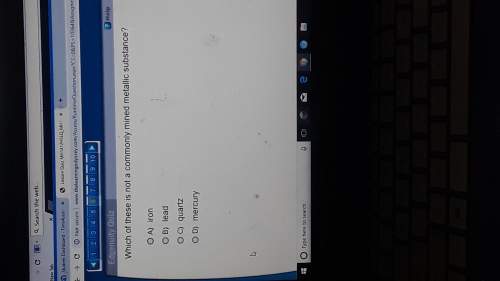
Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3
You know the right answer?
Protons + Neutrons = the atomic ?...
Questions

Mathematics, 17.09.2020 06:01

Physics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Physics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Biology, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

History, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01

Mathematics, 17.09.2020 06:01