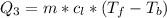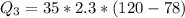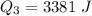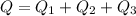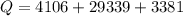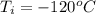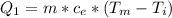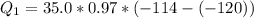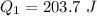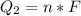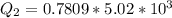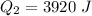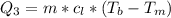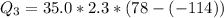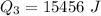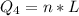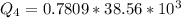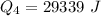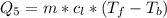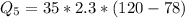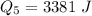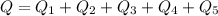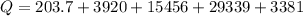Ethanol (C2H5OH) melts at –114 °C and boils at 78 °C. The enthalpy of fusion of ethanol is 5.02 kJ/mol, and its enthalpy of vaporization is 38.56 kJ/mol. The specific heats of solid and liquid ethanol are 0.97 J/g-K and 2.3 J/g-K, respectively. The average specific heat of gaseous ethanol is about 1.80 J/g-K. a. How much heat is required to convert 35.0 g of ethanol at 27 °C to the vapor phase at 120 °C? b. How much heat is required to convert the same amount of ethanol at –120 °C to the vapor phase at 120 °C?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2

You know the right answer?
Ethanol (C2H5OH) melts at –114 °C and boils at 78 °C. The enthalpy of fusion of ethanol is 5.02 kJ/m...
Questions

History, 08.04.2020 20:32





Mathematics, 08.04.2020 20:32


Mathematics, 08.04.2020 20:32

Mathematics, 08.04.2020 20:32






Mathematics, 08.04.2020 20:32

Mathematics, 08.04.2020 20:32

Mathematics, 08.04.2020 20:32

Mathematics, 08.04.2020 20:32

English, 08.04.2020 20:32

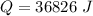
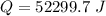
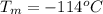
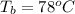
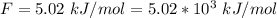
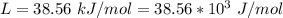
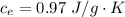
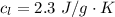
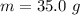
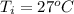
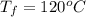
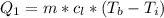
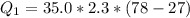
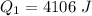
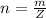
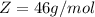
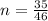
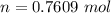
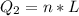
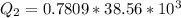
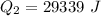
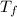 is mathematically represented as
is mathematically represented as