Chemistry, 03.12.2020 14:20 babyduckies37
25.0cm3 of s saturated potassium hydroxide is neutralized by 35.0cm3 of hydrogen chloride acid of concentration 0.75 mol/dm3. Calculate the concentration of potassium hydroxide solution. Please help, will give brainliest, unhelpful answers will get reported.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 10:30
Rocks, as they are compressed, begin forming mountains above the earth's surface when two continental plates converge. the continental crust increases in depth as the mountains grow above. the himalayan mountains formed at a convergent plate boundary in this manner. the rocks are smashed together causing them to due to the intense heat and pressure from the colliding plates and eventually forming rock. a) melt; igneous b) layer; sedimentary c) recrystallize; metamorphic d) melt into the earth's interior; metamorphic
Answers: 1

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3
You know the right answer?
25.0cm3 of s saturated potassium hydroxide is neutralized by 35.0cm3 of hydrogen chloride acid of co...
Questions

Mathematics, 16.11.2020 19:20

Mathematics, 16.11.2020 19:20


Mathematics, 16.11.2020 19:20



Mathematics, 16.11.2020 19:20



Mathematics, 16.11.2020 19:20


Mathematics, 16.11.2020 19:20


Business, 16.11.2020 19:20

Mathematics, 16.11.2020 19:20

Mathematics, 16.11.2020 19:20

Mathematics, 16.11.2020 19:20


Biology, 16.11.2020 19:20

History, 16.11.2020 19:20

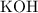 solution: approximately
solution: approximately 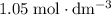 .
. 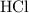 solution is in the unit
solution is in the unit 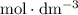 . However, the unit of the two volumes is
. However, the unit of the two volumes is 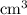 . Convert the unit of the two volumes to
. Convert the unit of the two volumes to 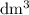 to match the unit of concentration.
to match the unit of concentration.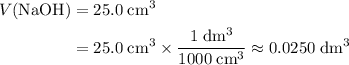 .
.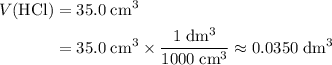 .
.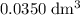 of
of 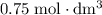
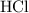 solution:
solution: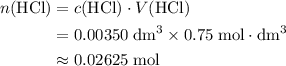 .
.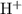 .
.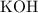 formula unit would react with up to one
formula unit would react with up to one 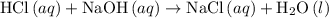 .
.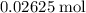 of
of 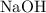 formula units. That is:
formula units. That is: 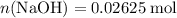 .
.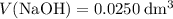 and
and 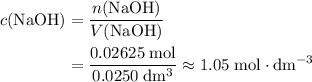 .
.