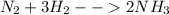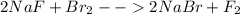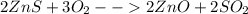Balance the following equations:
i. N2 + …..H2 → ….. NH3
ii. P4 + …….O2 → ….P2O5
iii. ……N...


Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
How many liters of water vapor can be produced if 108 grams of methane gas (ch4) are combusted at 312 k and 0.98 atm? show all work. pls ! will mark as brainliest
Answers: 1

Chemistry, 22.06.2019 02:10
Determine the percent sulfuric acid by mass of a 1.61 m aqueous solution of h2so4. %
Answers: 2

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2
You know the right answer?
Questions



Mathematics, 15.10.2020 23:01

Social Studies, 15.10.2020 23:01





Mathematics, 15.10.2020 23:01


English, 15.10.2020 23:01


Business, 15.10.2020 23:01


Mathematics, 15.10.2020 23:01

Social Studies, 15.10.2020 23:01


Chemistry, 15.10.2020 23:01


History, 15.10.2020 23:01