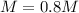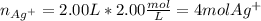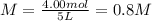Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3

Chemistry, 23.06.2019 01:30
Which statement justifies that hydrogen peroxide (h2o2) is a polar molecule? the o – h bond is nonpolar and the molecule is asymmetric. the o – h bond is nonpolar and the molecule is symmetric. the o – h bond is polar and the molecule is asymmetric. the o – h bond is polar and the molecule is symmetric.
Answers: 1

Chemistry, 23.06.2019 07:20
Which of the following are acids or bases? 1. sodium hydrogen 2. barium hydroxide solution 3. carbonate solution
Answers: 1

You know the right answer?
You have 3.00 L of a 3.12 M solution of NaCl(aq) called solution A. You also have 2.00 L of a 2.00 M...
Questions


Mathematics, 30.08.2019 07:10





History, 30.08.2019 07:10



Mathematics, 30.08.2019 07:10

Mathematics, 30.08.2019 07:10

Mathematics, 30.08.2019 07:10



Mathematics, 30.08.2019 07:10

Mathematics, 30.08.2019 07:10


Physics, 30.08.2019 07:10