
Chemistry, 02.12.2020 17:10 sabrinamarie17
Assume that the total volume of a metal sample is the sum of the volume occupied by the metal ions making up the lattice and the (separate) volume occupied by the conduction electrons. The density and molar mass of the first metal are 911 kg/m3 and 27.7 g/mol, respectively; assume the radius of an ion is 97.3 pm. (a) What percent of the volume of a sample of this metal is occupied by its conduction electrons

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Agas is contained in a thick walled balloon when the pressure changes from 1.21 atm to 2.52 the volume changes from 3.75 l to 1.72 l and the temperature change from 293k to blank k
Answers: 3


Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2
You know the right answer?
Assume that the total volume of a metal sample is the sum of the volume occupied by the metal ions m...
Questions

Mathematics, 22.08.2019 09:30

Physics, 22.08.2019 09:30



Physics, 22.08.2019 09:30

Mathematics, 22.08.2019 09:30

Social Studies, 22.08.2019 09:30






English, 22.08.2019 09:30

Health, 22.08.2019 09:30

Business, 22.08.2019 09:30





Social Studies, 22.08.2019 09:30

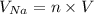
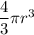
![V = \bigg [ \dfrac{4}{3} \pi r^3 \bigg ] \bigg [\dfrac{MN_A}{m} \bigg ]](/tpl/images/0941/6150/e576e.png)
![V = \bigg [ \dfrac{4}{3} \pi (97.3 \times 10^{-12} )^3 \bigg ] \bigg [\dfrac{ (911 kg/m^3 (m^3))(6.023\times 10^{23})}{27.7 \times10^{-3}\ kg/mol } \bigg ]](/tpl/images/0941/6150/33009.png)


