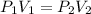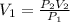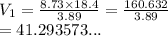Chemistry, 02.12.2020 08:50 whortonraijan
The pressure of a sample of argon gas was increased from 3.89 atm to 8.73 atm at constant temperature. If the final volume of the argon sample was 18.4 L, what was the initial volume of the argon sample? Assume ideal behavior.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 14:00
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2

Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1
You know the right answer?
The pressure of a sample of argon gas was increased from 3.89 atm to 8.73 atm at constant temperatur...
Questions



History, 07.08.2019 01:10




Mathematics, 07.08.2019 01:10


English, 07.08.2019 01:10

Mathematics, 07.08.2019 01:10

Mathematics, 07.08.2019 01:10

Mathematics, 07.08.2019 01:10

Physics, 07.08.2019 01:10


Spanish, 07.08.2019 01:10


Mathematics, 07.08.2019 01:10

Mathematics, 07.08.2019 01:10


Mathematics, 07.08.2019 01:10