1. Calculate the mass of iron needed to make 16.25g of iron chloride (3
marks)
2Fe + 3Cl...

Chemistry, 29.11.2020 18:10 emilykmorphew
1. Calculate the mass of iron needed to make 16.25g of iron chloride (3
marks)
2Fe + 3Cl2 → 2FeCl3

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2


Chemistry, 23.06.2019 09:00
A0.10 m aqueous solution of sodium sulfate is a better conductor of electricity than a 0.10 m aqueous solution of sodium chloride. which of the following best explains this observation? (a) sodium sulfate is more soluble in water than sodium chloride. (b) sodium sulfate has a higher molar mass than sodium chloride. (c) to prepare a given volume of 0.10 m solution, the mass of sodium sulfate needed is more than twice the mass of sodium chloride needed. (d) more moles of ions are present in a given volume of 0.10 m sodium sulfate than in the same volume of 0.10 m sodium chloride. (e) the degree of dissociation of sodium sulfate in solution is significantly greater than that of sodium chloride.
Answers: 2
You know the right answer?
Questions

Computers and Technology, 29.07.2019 21:10













Mathematics, 29.07.2019 21:20

Mathematics, 29.07.2019 21:20


Biology, 29.07.2019 21:20

Mathematics, 29.07.2019 21:20

Chemistry, 29.07.2019 21:20

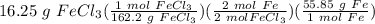 = 5.59533 g Fe
= 5.59533 g Fe

