
Chemistry, 28.11.2020 05:30 kingalbyss1230
Calculate how many grams of sodium hypochlorite can be theoretically formed when 1.23 mol of sodium hydroxide is combined with 1.36 mol of chlorine.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2

Chemistry, 23.06.2019 06:50
The student repeated the experiment using a higher concentration of acid. the same volume of acid and the same mass of magnesium ribbon were used. what volume of hydrogen gas would have been produced after 60 seconds?
Answers: 1
You know the right answer?
Calculate how many grams of sodium hypochlorite can be theoretically formed when 1.23 mol of sodium...
Questions


Mathematics, 06.09.2019 02:30

Mathematics, 06.09.2019 02:30





History, 06.09.2019 02:30

Biology, 06.09.2019 02:30



Mathematics, 06.09.2019 02:30


Mathematics, 06.09.2019 02:30


Social Studies, 06.09.2019 02:30




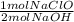 = 0.615 mol NaClO
= 0.615 mol NaClO

