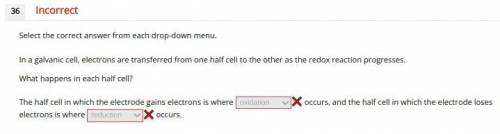
Chemistry, 26.11.2020 23:20 VICTURN3666
Identify the term that matches each electrochemistry definition.
a. An electrochemical cell powered by a spontaneous redox reaction.
b. The electrode where reduction occurs.
c. The electrode where oxidation occurs
d. An electrochemical cell that takes in energy to carry out a nonspontaneous redox reaction.
e. A chemical equation showing either oxidation or reduction.
1. Cathode
2. Anode
3. Galvanic cell
4. Half reaction
5. Electrolytic cell

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Diamond, graphite, and fullerenes share what property? a. they are all made of carbon (c) bonded to a metal. b. their shape. c. they are all made of carbon (c). d. they are all good conductors.
Answers: 1

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

You know the right answer?
Identify the term that matches each electrochemistry definition.
a. An electrochemical cell powered...
Questions






Biology, 06.05.2020 09:04

Social Studies, 06.05.2020 09:04

Mathematics, 06.05.2020 09:04


Mathematics, 06.05.2020 09:04

History, 06.05.2020 09:04



Mathematics, 06.05.2020 09:04




Mathematics, 06.05.2020 09:04

Mathematics, 06.05.2020 09:04