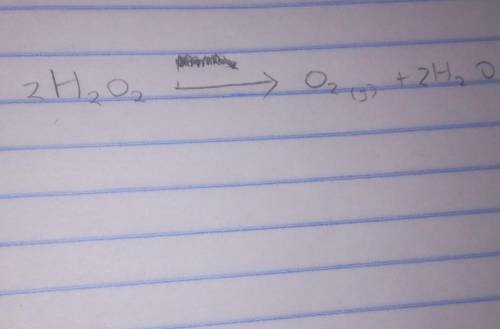
1. Lauri conducts an experiment to investigate how the mass of a catalyst, manganese (IV) oxide, affects the rate of production of oxygen during the decomposition of hydrogen peroxide (H2O2). Water and heat are also produced during the decomposition.
(a) (i) Name TWO factors OTHER THAN a catalyst, which can affect the rate of a chemical reaction. (2mks)
(ii) Write a balanced equation to show the decomposition of hydrogen peroxide by manganese (IV) oxide, MnO2(s) (2mks)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 23.06.2019 00:50
Which of the following warnings would an agricultural chemist tell a farmer who wants to recycle his or her own ammonia? recycling ammonia is a difficult process that sometimes takes weeks. recycling ammonia requires a degree in biochemistry or a related field. recycling ammonia can be harmful because it is highly flammable and toxic. recycling ammonia costs too much money considering the price of the necessary chemicals.
Answers: 1

You know the right answer?
1. Lauri conducts an experiment to investigate how the mass of a catalyst, manganese (IV) oxide, aff...
Questions

Mathematics, 31.01.2020 17:01

Mathematics, 31.01.2020 17:01



History, 31.01.2020 17:01

Social Studies, 31.01.2020 17:01

Mathematics, 31.01.2020 17:01

History, 31.01.2020 17:01



Mathematics, 31.01.2020 17:01

Physics, 31.01.2020 17:01

Mathematics, 31.01.2020 17:01





Mathematics, 31.01.2020 17:01