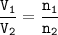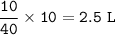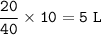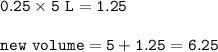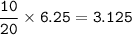The chemical equilibrium is given:
2NO (g) + O2(g) <—>2N O2, (g)
In a container...

Chemistry, 25.11.2020 14:00 shartman22
The chemical equilibrium is given:
2NO (g) + O2(g) <—>2N O2, (g)
In a container of volume 10L is in equilibrium a mixture consisting of 10 mol NO (g), 10 mol O2, (g) and 20 mol NO2, (g).
The volume of the container changes under constant temperature and after the restoration of equilibrium the amount of NO has increased by 25%. Calculate the volume change in L.
How do I find the change in volume?
P. s sorry if my English isn’t perfect.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
The skeletal system performs a variety of functions that are crucial to maintaining life processes. what function is performed in the bone marrow, but not in the ossified bones of the skeleton? a oxygen transportation c mineral storage b. muscle attachment d red blood cell production
Answers: 3


Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

You know the right answer?
Questions




Biology, 02.08.2019 16:00



English, 02.08.2019 16:00


Biology, 02.08.2019 16:00


English, 02.08.2019 16:00


Social Studies, 02.08.2019 16:00


History, 02.08.2019 16:00

Social Studies, 02.08.2019 16:00


Mathematics, 02.08.2019 16:00