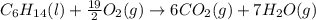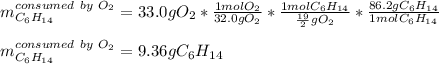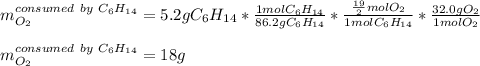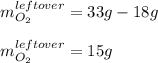Liquid hexane CH3CH24CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gaseous water H2O. Suppose 5.2 g of hexane is mixed with 33.0 g of oxygen. Calculate the minimum mass of hexane that could be left over by the chemical reaction. Be sure your answer has the correct number of significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
What is the ph of a solution with a concentration of 5.2 × 10–8 m h3o+?
Answers: 1

Chemistry, 21.06.2019 16:00
Based on the law of conservation of energy, which statement is false? answer- energy is lost when machines dont work right
Answers: 1

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1
You know the right answer?
Liquid hexane CH3CH24CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and...
Questions

Mathematics, 11.01.2021 04:50




Mathematics, 11.01.2021 05:00

Arts, 11.01.2021 05:00

Biology, 11.01.2021 05:00


English, 11.01.2021 05:00


Mathematics, 11.01.2021 05:00

English, 11.01.2021 05:00

Mathematics, 11.01.2021 05:00

Chemistry, 11.01.2021 05:00


Physics, 11.01.2021 05:00

Social Studies, 11.01.2021 05:00

Physics, 11.01.2021 05:00


Geography, 11.01.2021 05:00