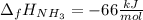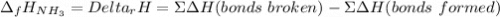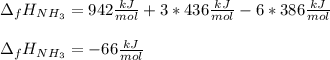Chemistry, 23.11.2020 02:30 afitzgerald
The formation of ammonia is represented by the equation N2(g) + 3H2(g) ⇌ 2NH3(g). Determine the enthalpy of formation of ammonia given the following mean bond enthalpies (kJmol-1): N≡N 942; H-H 436; N-H 386

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Chemistry, 22.06.2019 05:30
What royal scientist used the 29th day of frozen vapor to encounter elements for mastering new culinary creations?
Answers: 1


Chemistry, 22.06.2019 22:30
What is the value of the standard enthalpy of formation of an element in its most stable form?
Answers: 3
You know the right answer?
The formation of ammonia is represented by the equation N2(g) + 3H2(g) ⇌ 2NH3(g). Determine the enth...
Questions

Mathematics, 12.05.2020 19:57


History, 12.05.2020 19:57

Mathematics, 12.05.2020 19:57

Mathematics, 12.05.2020 19:57


Mathematics, 12.05.2020 19:57


Mathematics, 12.05.2020 19:57






Social Studies, 12.05.2020 19:57

Mathematics, 12.05.2020 19:57


Physics, 12.05.2020 19:57

Mathematics, 12.05.2020 19:57

Geography, 12.05.2020 19:57