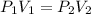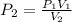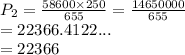Chemistry, 23.11.2020 01:00 Homepage10
A sample of air at 58.6 kPa is expanded from 250 mL to 655 mL. If thetemperature remains constant, what is the final pressure in kPa?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2

Chemistry, 23.06.2019 09:00
Astream of surface water reaches a porous portion of sediment and seeps into the ground. this water eventually joins a large reservoir of water located beneath the earth's surface. the example above describes the interacting with the a. cryosphere; biosphere b. hydrosphere; biosphere c. hydrosphere; geosphere d. cryosphere; geosphere
Answers: 3
You know the right answer?
A sample of air at 58.6 kPa is expanded from 250 mL to 655 mL. If thetemperature remains constant, w...
Questions






History, 30.07.2019 05:00

English, 30.07.2019 05:00





Physics, 30.07.2019 05:00




Physics, 30.07.2019 05:00




Mathematics, 30.07.2019 05:00