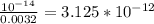Chemistry, 20.11.2020 14:00 sunshine52577oyeor9
Calculate the pH of the resulting solution if 32.0 mL of 0.320 M HCl(aq) is added to (a) 42.0 mL of 0.320 M NaOH(aq). (b) 22.0 mL of 0.420 M NaOH(aq).

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Which term best describes the form sound takes as it travels away from a drum (a- gas)(b-music) ( c-waves) (d-particles
Answers: 3

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2


Chemistry, 23.06.2019 05:30
Scientist think that animals with remarkably similar embryo development probably shared a common ancestor
Answers: 1
You know the right answer?
Calculate the pH of the resulting solution if 32.0 mL of 0.320 M HCl(aq) is added to (a) 42.0 mL of...
Questions

Mathematics, 22.09.2019 01:30

English, 22.09.2019 01:30

Biology, 22.09.2019 01:30

Chemistry, 22.09.2019 01:30

Mathematics, 22.09.2019 01:30


Biology, 22.09.2019 01:30





Mathematics, 22.09.2019 01:30

Physics, 22.09.2019 01:30

English, 22.09.2019 01:30


Computers and Technology, 22.09.2019 01:30

Business, 22.09.2019 01:30

Social Studies, 22.09.2019 01:30

History, 22.09.2019 01:30

Chemistry, 22.09.2019 01:30