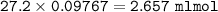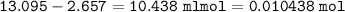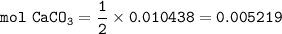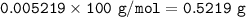Chemistry, 18.11.2020 21:30 lilpeepxliltracy
1.3188g of antacid is weighed and mixed with 75.00 mL of excess 0.1746 M HCl. The excess wcid required 27.20 mL of 0.09767 M NaOH for back titration. Calculate the amount of CaCO3 in the tablet.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:40
If the atomic mass of an atom is 34 and the atom contains 13 protons, how many neutrons does the atom contain?
Answers: 2

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2

Chemistry, 22.06.2019 22:30
Astudent pours 10.0 g of salt into a container of water and observes the amount of time it takes for the salt to dissolve. she then repeats the process using the same amounts of salt and water but this time she slowly stirs the mixture while it is dissolving. the student performs the experiment one more time but this time she stirs the mixture rapidly. the dependent variable in this experiment is: time for salt to dissolve speed of stirring amount of water mass of salt
Answers: 1
You know the right answer?
1.3188g of antacid is weighed and mixed with 75.00 mL of excess 0.1746 M HCl. The excess wcid requir...
Questions


Mathematics, 29.04.2021 23:00

Mathematics, 29.04.2021 23:00

Social Studies, 29.04.2021 23:00

History, 29.04.2021 23:00


Biology, 29.04.2021 23:00


Mathematics, 29.04.2021 23:00



Mathematics, 29.04.2021 23:00


Mathematics, 29.04.2021 23:00

Mathematics, 29.04.2021 23:00

Mathematics, 29.04.2021 23:00


Mathematics, 29.04.2021 23:00

Spanish, 29.04.2021 23:00