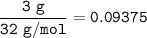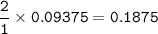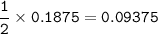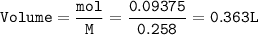Chemistry, 15.11.2020 23:10 scadengo123
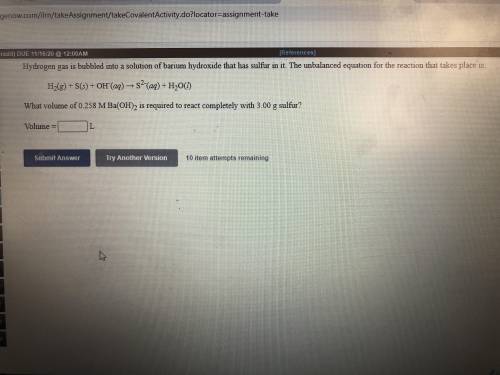
Hydrogen gas is bubbled into a solution of barium hydroxide that has sulfur in it. The unbalanced equation for the reaction that takes place is: H2+S+OH- —-> S2- + H2O What volume of 0.258 M Ba(OH)2 is required to react completely with 3.00g sulfur?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:10
Agas mixture with a total pressure of 745 mmhg contains each of the following gases at the indicated partial pressures: co2, 245 mmhg ; ar, 119 mmhg ; and o2, 163 mmhg . the mixture also contains helium gas. part a what is the partial pressure of the helium gas? phe p h e = nothing mmhg request answer part b what mass of helium gas is present in a 10.2-l sample of this mixture at 283 k ? m m = nothing g request answer
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2
You know the right answer?
Hydrogen gas is bubbled into a solution of barium hydroxide that has sulfur in it. The unbalanced eq...
Questions

Mathematics, 26.04.2021 04:10


Social Studies, 26.04.2021 04:10

Mathematics, 26.04.2021 04:10


Advanced Placement (AP), 26.04.2021 04:10



Mathematics, 26.04.2021 04:10

Computers and Technology, 26.04.2021 04:10

Mathematics, 26.04.2021 04:10


History, 26.04.2021 04:10

Mathematics, 26.04.2021 04:10


Spanish, 26.04.2021 04:10